GM medicinal products: Regulatory procedures
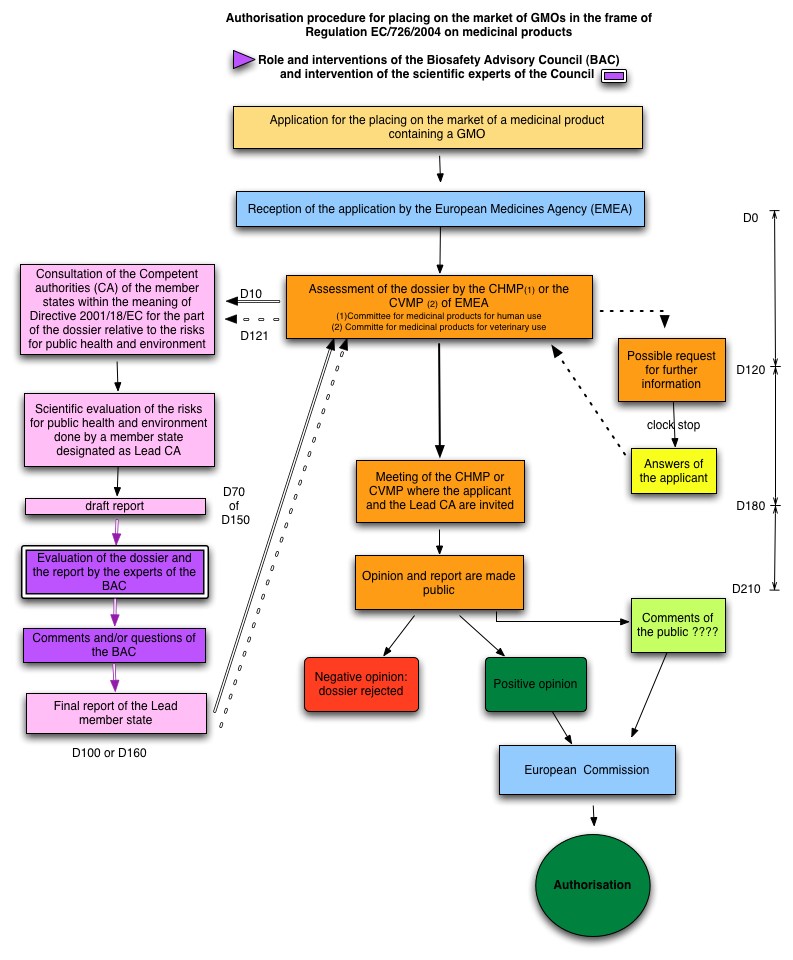
The roles and interventions of the Council in the regulatory procedure for notifications submitted for the placing on the market of a medicinal product containing a GMO dossiers under Regulation (EC) 726/2004 are summarised in the diagram on the right side.
For the scientific evaluation of these dossiers and in addition to the abovementioned Regulation, the Council can refer to several guidelines, listed on the Belgian Biosafety Server.